Free Stock Trading is Just the Beginning
Free stock trading and Free trading software
In addition to commission free stock trading, TradeZero provides free limit orders, higher day trading leverage, 24 x 7 customer support along with four different state-of-the-art trading software and more.
Learn MoreFree Online Stock Trading Software
Access your online stock trading account wherever you go with ZeroMobile.
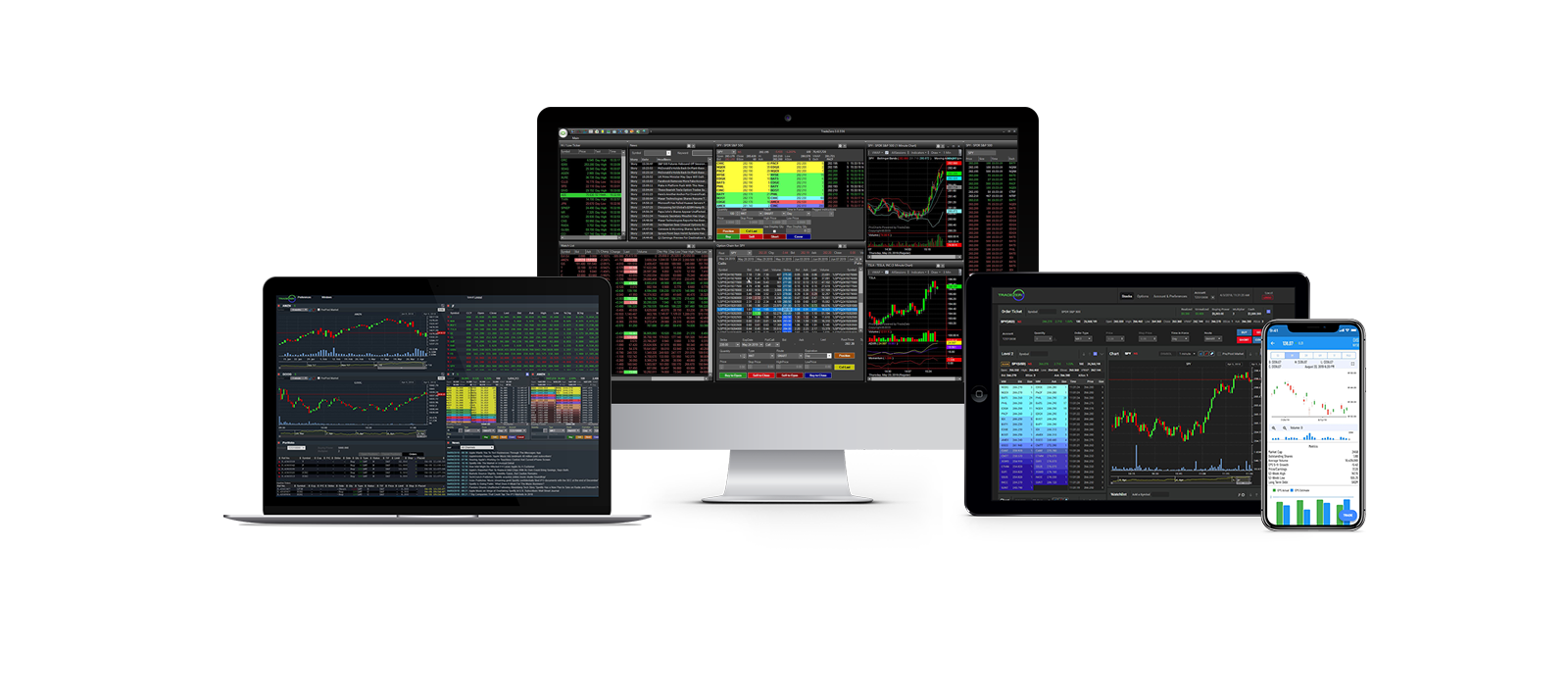
ZeroPro
ZeroPro provides the speed and all the features that are needed for active traders.
Why Choose TradeZero Access your trading account wherever you go
 24 x 7Customer Support.
24 x 7Customer Support.
Call anytime from 7am to 5pm ET or start a chat on the website 24 x 7. Your choice...
Learn More Free Limit Orders
Free Limit Orders
We provide 100% free limit orders to all of our accounts. Open an Account with TradeZero...
Learn More Receive upto 6 to 1 Leverage
Receive upto 6 to 1 Leverage
TradeZero provides clients up to 6 to 1 intraday leverage on their equity....
Learn More Our Technology
Our Technology
TradeZero offers all clients state-of-the-art trading software. Whether you're a beginner...
Learn More No Pattern Day Trading Rules
No Pattern Day Trading Rules
No Pattern Day Trading Rules. We're the best brokerage for pattern day traders. No pattern...
Learn MoreReward Based Partnership
Our partnership program rewards individuals and companies in the trading community. If you have an audience, simply refer your clients to your customized TradeZero URL. We'll also add you to our Partnership Landing page to benefit from our TradeZero Community.
What People Say
Register for a Free Software Demo
Open and fund a trading account today and experience The TradeZero Advantage.

